Trusted for 100% GxP Compliance for Over 30 Years
ELPRO experts can qualify and maintain your systems, equipment, rooms or facilities through a wide range of GxP services. Schedule a project scoping call today.
ELPRO experts can qualify and maintain your systems, equipment, rooms or facilities through a wide range of GxP services. Schedule a project scoping call today.
ELPRO experts can qualify and maintain your systems, equipment, rooms or facilities through a wide range of GxP services. Schedule a project scoping call today.
ELPRO facilitated the qualification of both the temperature monitoring equipment as well as our cold storage units by providing excellent expertise and technical support just in time.
We had a really fruitful collaboration with ELPRO concerning our Qualification and mapping of our new GMP/GDP warehouse. All steps until the authority inspection were attended professionally and finally we had no issues.
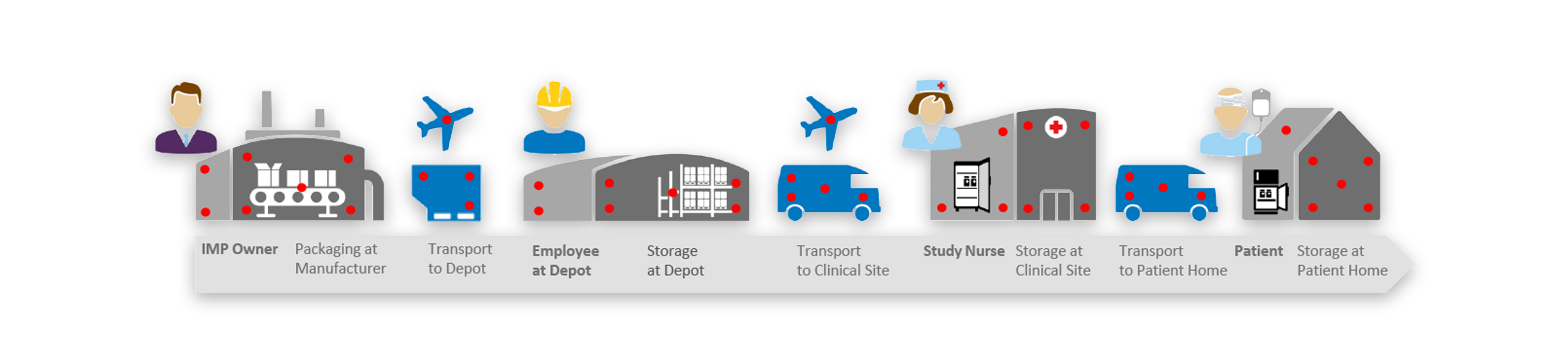
Complete the form below to tell us a little more about up upcoming project and we’ll have one of our GxP services professionals contact you to set up a project scoping call and gather all the details. We look forward to speaking with you soon.
we prove it.

Copyright © ELPRO-BUCHS AG 2024. All Rights Reserved.